-
About
-
Academics
- Physician Assistant
- Special Master’s (MBS)
-
Admissions & Financial Aid
- Tuition & Fees
-
Student Life
-
Research
- Research Labs & Centers
-
Local & Global Engagement
- Global Health Program
What Parents Need to Know About the COVID-19 Vaccine for Kids
A Tufts pediatrics expert explains how vaccine dosages for children are developed and why it’s important for young people to be vaccinated
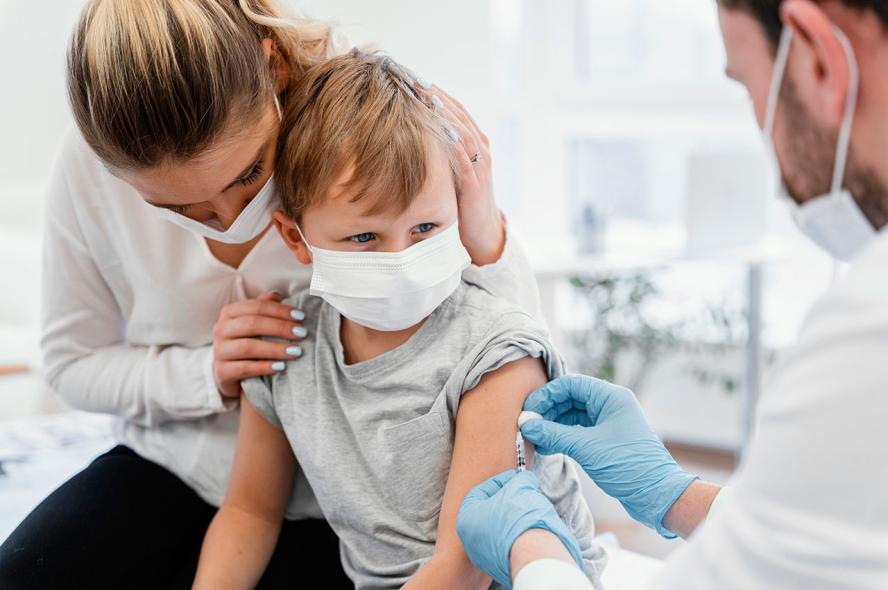
On October 29, the U.S. Food and Drug Administration (FDA) gave emergency authorization for a child-size dose of Pfizer’s COVID-19 vaccine for use in kids ages 5 to 11. Some 28 million children could be eligible by early November for the two-shot regimen administered three weeks apart.
Daniel Rauch, a professor of pediatrics at Tufts University School of Medicine and chief of pediatric hospital medicine at Tufts Medical Center, spoke with Tufts Now about how vaccines get developed for children and why he will recommend this one.